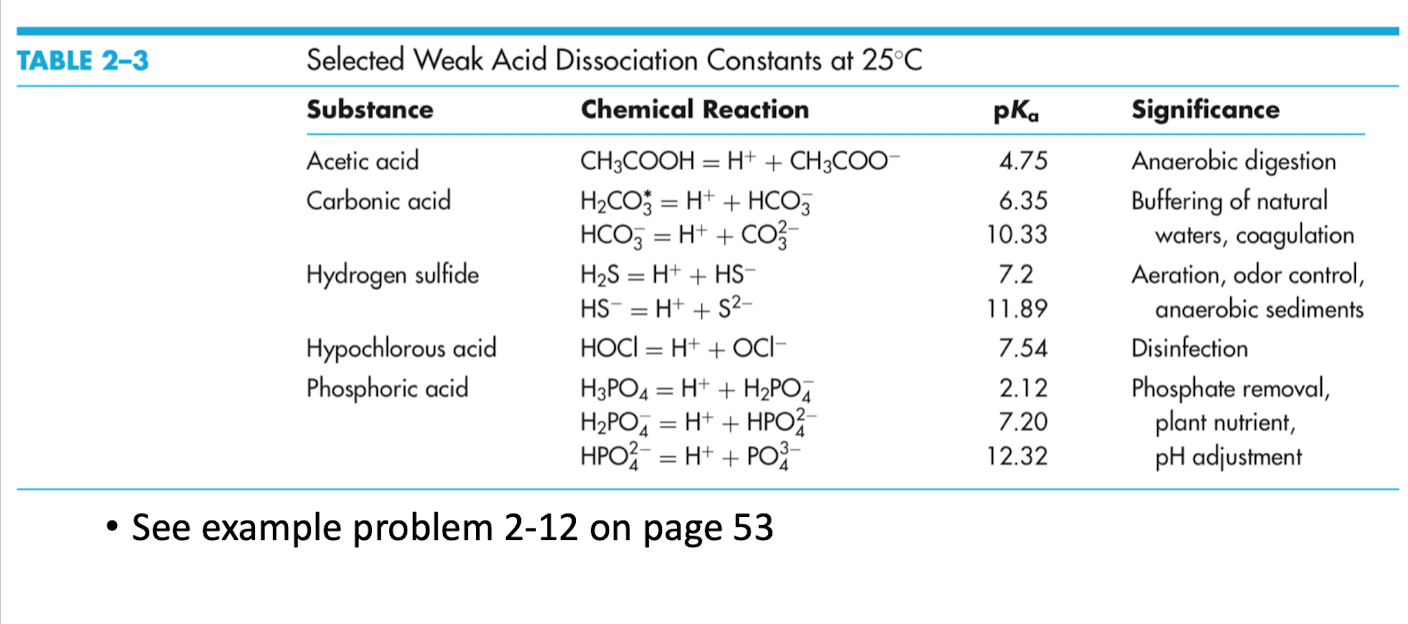
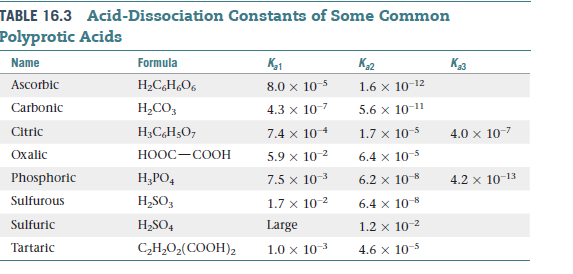
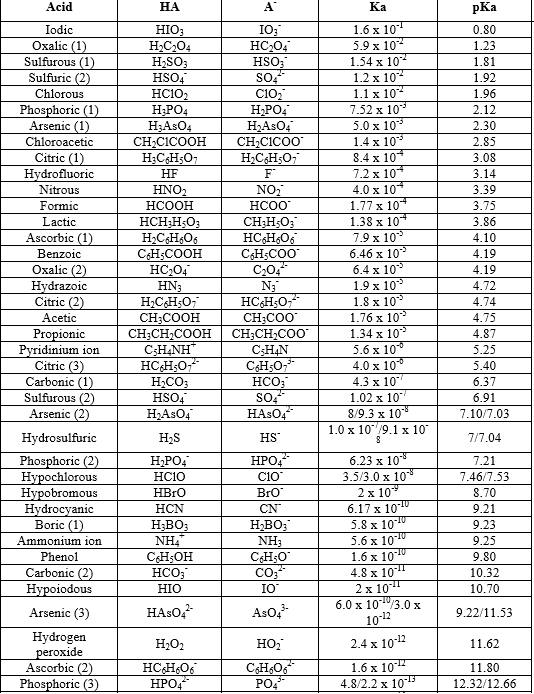
![PDF] Acid Dissociation Constants and Related Thermodynamic Functions of Protonated 2,2-Bis(Hydroxymethyl)-2,2',2”- Nitrilotriethanol (BIS-TRIS) from (278.15 to 328.15) K | Semantic Scholar PDF] Acid Dissociation Constants and Related Thermodynamic Functions of Protonated 2,2-Bis(Hydroxymethyl)-2,2',2”- Nitrilotriethanol (BIS-TRIS) from (278.15 to 328.15) K | Semantic Scholar](https://d3i71xaburhd42.cloudfront.net/b10c6e789b338b6488dc7e3e0f9701647bb9b281/4-Table2-1.png)
PDF] Acid Dissociation Constants and Related Thermodynamic Functions of Protonated 2,2-Bis(Hydroxymethyl)-2,2',2”- Nitrilotriethanol (BIS-TRIS) from (278.15 to 328.15) K | Semantic Scholar
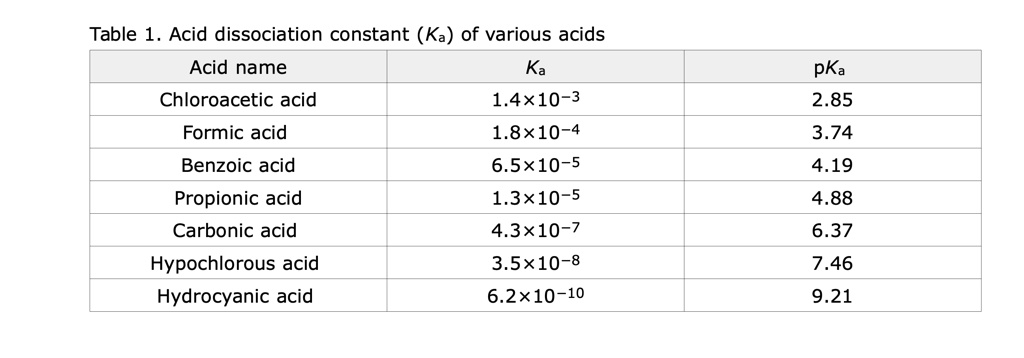
SOLVED: Acid dissociation constant (Ka) of various acids: Acid name Ka Chloroacetic acid 1.4x10^-3 Formic acid 1.8x10^-4 Benzoic acid 6.5x10^-5 Propionic acid 1.3x10^-5 Carbonic acid 4.3x10^-7 Hypochlorous acid 3.5x10^-8 Hydrocyanic acid 6.2x10^-10
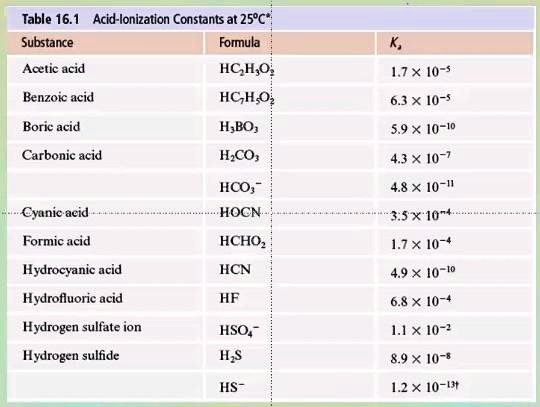
SOLVED: Table 16.1 Acid-Ionization Constants at 25°C Substance Formula Acid -Ionization Constant (x 10^-5) Acetic acid HC2H3O2 1.8 Benzoic acid C6H5COOH 6.3 Boric acid H3BO3 5.9 x 10^-10 Carbonic acid H2CO3 4.8 x

Table 2 from Measurement of dissociation constants (pKa values) of organic compounds by multiplexed capillary electrophoresis using aqueous and cosolvent buffers. | Semantic Scholar
![PDF] Determination of Dissociation Constants of Malonic Acid in (Ethylene Glycol-Water)X% Mixed Solvent at Different Temperatures Using Electromotive Force Measurements | Semantic Scholar PDF] Determination of Dissociation Constants of Malonic Acid in (Ethylene Glycol-Water)X% Mixed Solvent at Different Temperatures Using Electromotive Force Measurements | Semantic Scholar](https://d3i71xaburhd42.cloudfront.net/6d04a3862b2f2dbb01b9220329429f516e4430e9/2-Table1-1.png)
PDF] Determination of Dissociation Constants of Malonic Acid in (Ethylene Glycol-Water)X% Mixed Solvent at Different Temperatures Using Electromotive Force Measurements | Semantic Scholar
![PDF] THE APPARENT DISSOCIATION CONSTANTS OF ARGININE AND OF LYSINE AND THE APPARENT HEATS OF IONIZATION OF CERTAIN AMINO ACIDS | Semantic Scholar PDF] THE APPARENT DISSOCIATION CONSTANTS OF ARGININE AND OF LYSINE AND THE APPARENT HEATS OF IONIZATION OF CERTAIN AMINO ACIDS | Semantic Scholar](https://d3i71xaburhd42.cloudfront.net/fd5bc9e3adbccba2e9000c2716b5380a77442e08/7-TableII-1.png)
PDF] THE APPARENT DISSOCIATION CONSTANTS OF ARGININE AND OF LYSINE AND THE APPARENT HEATS OF IONIZATION OF CERTAIN AMINO ACIDS | Semantic Scholar

Table 1 from Determination of Acid Dissociation Constants (pKa) of Bicyclic Thiohydantoin-Pyrrolidine Compounds in 20% Ethanol-Water Hydroorganic Solvent | Semantic Scholar